Court Rules FDA Abused Authority With Anti-Ivermectin Messaging
In a landmark ruling on September 1, a federal court stated that the U.S. Food and Drug Administration (FDA) abused its authority under federal law when it advised Americans to “stop” using the drug Ivermectin as a treatment for COVID-19.
The Gateway Pundit previously reported that a group of doctors had filed a federal lawsuit against the U.S. Department of Health and Human Services (HHS) and the Food and Drug Administration (FDA) over the agencies’ unlawful attempts to block the use of Ivermectin in treating COVID-19.
The lawsuit, filed in the U.S. Southern District of Texas in Galveston, argues that the FDA has overstepped its authority and unjustifiably interfered with their medical practice.
During a hearing last year, the agency’s lawyers argued that the FDA was only giving advice and it was not mandatory when it told people to “stop” taking Ivermectin for COVID-19.
“The cited statements were not directives,” said Isaac Belfer, one of the lawyers. “They were not mandatory. They were recommendations. They said what parties should do. They said, for example, why you should not take Ivermectin to treat COVID-19. They did not say you may not do it, you must not do it. They did not say it’s prohibited or it’s unlawful. They also did not say that doctors may not prescribe Ivermectin.”
“They use informal language, that is true… It’s conversational but not mandatory,” he continued.
The statement from the lawyer contradicted the FDA’s social media post, stating, “Hold your horses, y’all. Ivermectin may be trending, but it still isn’t authorized or approved to treat COVID-19″ and another tweet says, “You are not a horse. You are not a cow. Seriously, y’all. Stop it.” Both posts were linked to FDA’s website with the title, “Why You Should Not Use Ivermectin to Treat or Prevent COVID-19.”
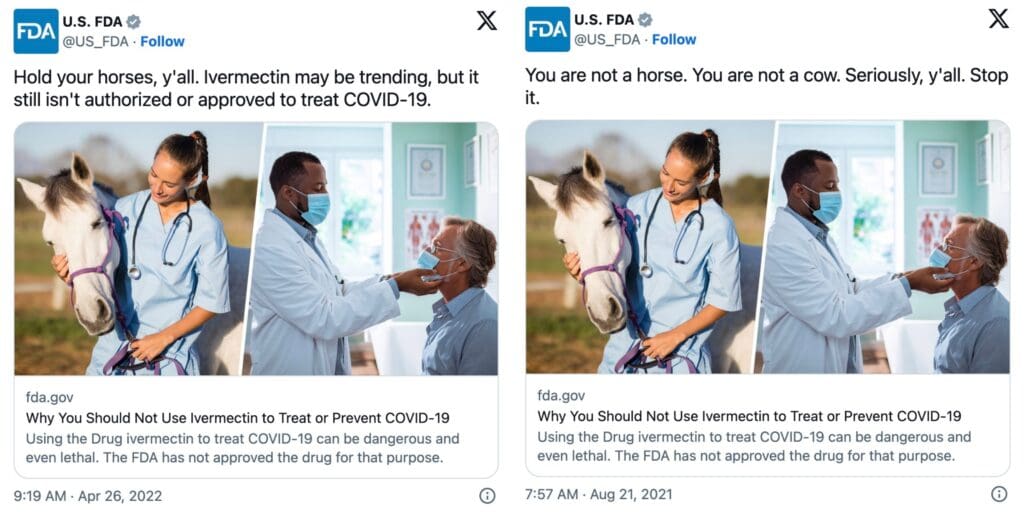
Last month, the lawyer representing the FDA clarified the agency’s stance on the use of Ivermectin for treating COVID-19 patients. The lawyer confirmed that doctors have the authority to prescribe the drug for off-label use in treating COVID-19.
During the oral argument, Ashley Cheung Honold, a Department of Justice lawyer representing the FDA stated that the agency “explicitly recognizes” that doctors do have the authority to administer Ivermectin to treat COVID.
“”FDA explicitly recognizes that doctors do have the authority to prescribe Ivermectin to treat COVID,” said Honold.
“FDA made these statements in response to multiple reports of consumers being hospitalized, after self-medicating with Ivermectin intended for horses, which is available for purchase over the counter without the need for prescription,” Honold said.
On Friday, September 1, U.S. Circuit Judge Don Willett stated in his written ruling, “FDA can inform, but it has identified no authority allowing it to recommend consumers ‘stop’ taking medicine.”
According to the court’s decision, the three physicians can now pursue legal action, arguing that the FDA’s campaign went beyond the scope of the agency’s jurisdiction as defined by federal legislation.
“FDA is not a physician. It has authority to inform, announce, and apprise—but not to endorse, denounce, or advise,” Judge Don Willett wrote for a panel that also included Jennifer Walker Elrod and Edith Brown Clement. “The Doctors have plausibly alleged that FDA’s Posts fell on the wrong side of the line between telling about and telling to. As such, the Doctors can use the APA to assert their ultra vires claims against the Agencies and the Officials.”
In the ruling, Judge Willett wrote, “Left unmentioned in most of that messaging: Ivermectin also comes in a human version. And while the human version of Ivermectin is not FDA-approved to treat the coronavirus, some people were using it off-label for that purpose.”
He added, “Even tweet-sized doses of personalized medical advice are beyond FDA’s statutory authority.”
In a statement to Epoch Times, Dr. Mary Talley Bowden said, “The FDA misled the public into thinking it has more authority than it does. This decision confirms that the FDA is not your doctor and has no authority to tell doctors how to practice medicine.”
It can be recalled that Houston Methodist launched an investigation into Bowden and suspended her for defying health authorities and exercising free speech.
The hospital excoriated Bowden for “using her social media accounts to express her personal opinions about the COVID-19 vaccine and treatments,” NBC News reports. The suspension barred the physician from admitting or treating patients at the hospital.
Bowden repeatedly warned that it is “wrong” to mandate the experimental mRNA vaccines and continuously touted Ivermectin as a safe and effective treatment amid threats from public health officials against prescribing the drug.
Bowden was forced to resign. In her resignation letter, Bowden doubled down on the efficacy of Ivermectin.
“I have worked hard to provide early treatment for victims of COVID-19. My efforts have been successful. I have treated more than 200 COVID-19 patients, including many with co-morbidities, and none of these patients have required hospitalization. This is a testament to the success of my treatment methods,” she wrote. “Throughout this pandemic, there has been no FDA-approved treatment for COVID. Therefore I have done my best to care for patients and save lives in the absence of a clear scientific consensus.”
“Early treatment must still be part of any strategy for patient care. That is why physicians and hospitals should pay more attention to medications such as Ivermectin, which significant research and my clinical experience indicate is effective,” she continued. “I have decided to part ways with Houston Methodist because of the accusation that I have been spreading “dangerous information.” This is false and defamatory. I do not spread misinformation, and my opinions are supported by science. There is substantial evidence for the efficacy of Ivermectin in treating COVID-19, and no evidence for serious or fatal side effects associated with the doses used to treat COVID-19.”
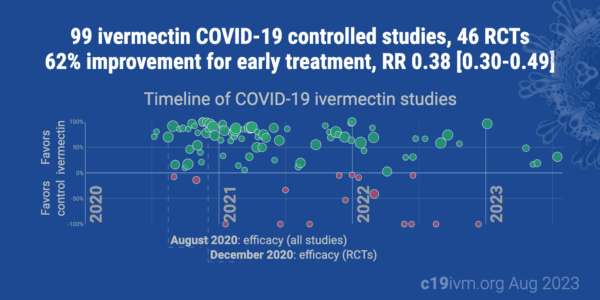
There have now been 99 Ivermectin COVID-19 controlled studies that show a 62% improvement in COVID patients.
Source: https://www.thegatewaypundit.com/2023/09/federal-court-rules-fda-abused-its-authority-anti




